
As home to a quarter of the world's known thorium reserves and notably lacking in uranium resources, it's no surprise that India envisions meeting 30% of its electricity demand through thorium-based reactors by 2050. Researchers have studied thorium-based fuel cycles for 50 years, but India leads the pack when it comes to commercialization. Thorium nuclear waste only stays radioactive for 500 years, instead of 10,000, and there is 1,000 to 10,000 times less of it to start with. With thorium, the U233 is isolated and the result is far fewer highly radioactive, long-lived byproducts. 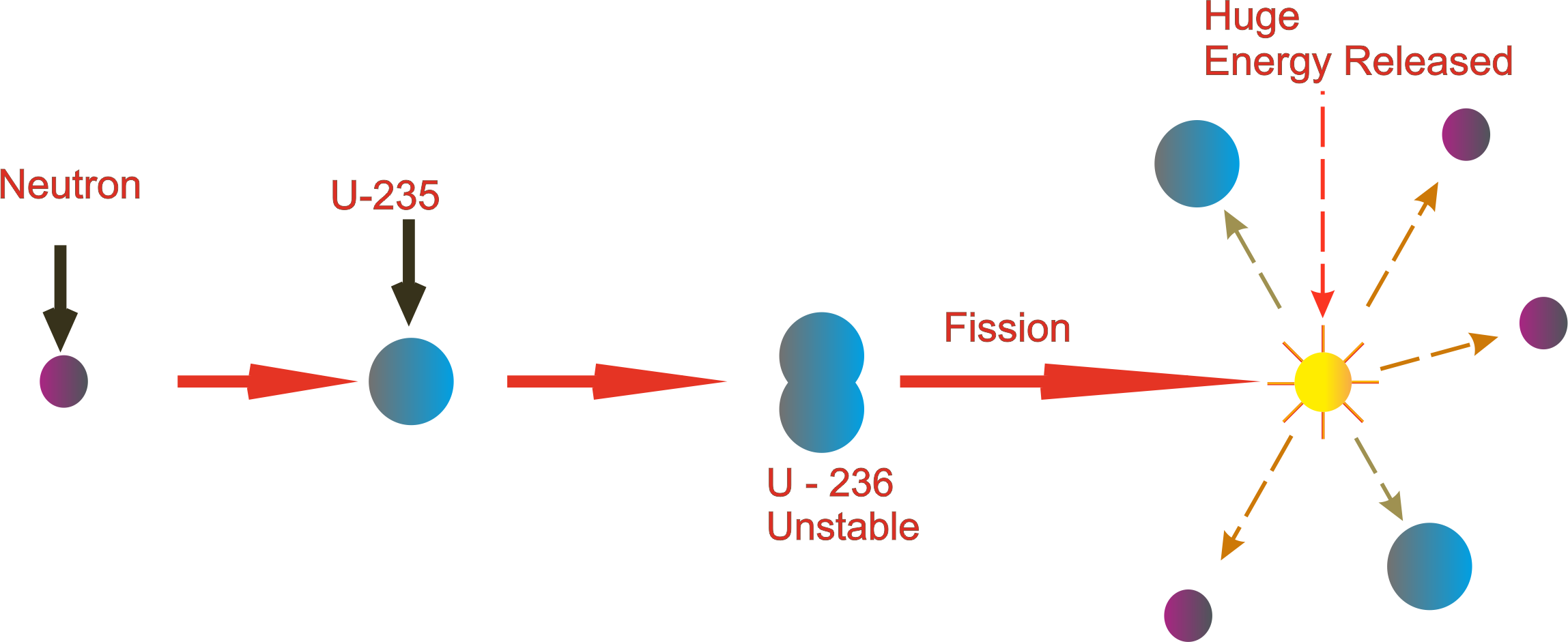
Remember, with uranium reactors it's the U238, turned into U239 by absorbing some of those high-flying neutrons, that produces all the highly radioactive waste products. But there isn't a pile of U238 sitting by. The U233 does its thing, splitting apart and releasing high-energy neutrons. The uranium is then fed into another reactor all on its own, to generate energy. Thankfully, it is also relatively long lived, which means at this point in the cycle the irradiated fuel can be unloaded from the reactor and the U233 separated from the remaining thorium. That's the same uranium isotope we use in reactors now as a nuclear fuel, the one that is fissile all on its own. When Th232 absorbs a neutron it becomes Th233, which is unstable and decays into protactinium-233 and then into U233. Then, when you need the reaction to stop, simply turn off the source of neutrons and the whole process shuts down, simple as pie. If you want to make thorium nuclei split apart, though, it's easy: you simply start throwing neutrons at them. That means no matter how many thorium nuclei you pack together, they will not on their own start splitting apart and exploding. Then there's the safety side of thorium reactions. That's a heck of a lot better than the 3% to 5% of uranium that comes in the form we need. Thorium's advantages start from the moment it is mined and purified, in that all but a trace of naturally occurring thorium is Th232, the isotope useful in nuclear reactors. 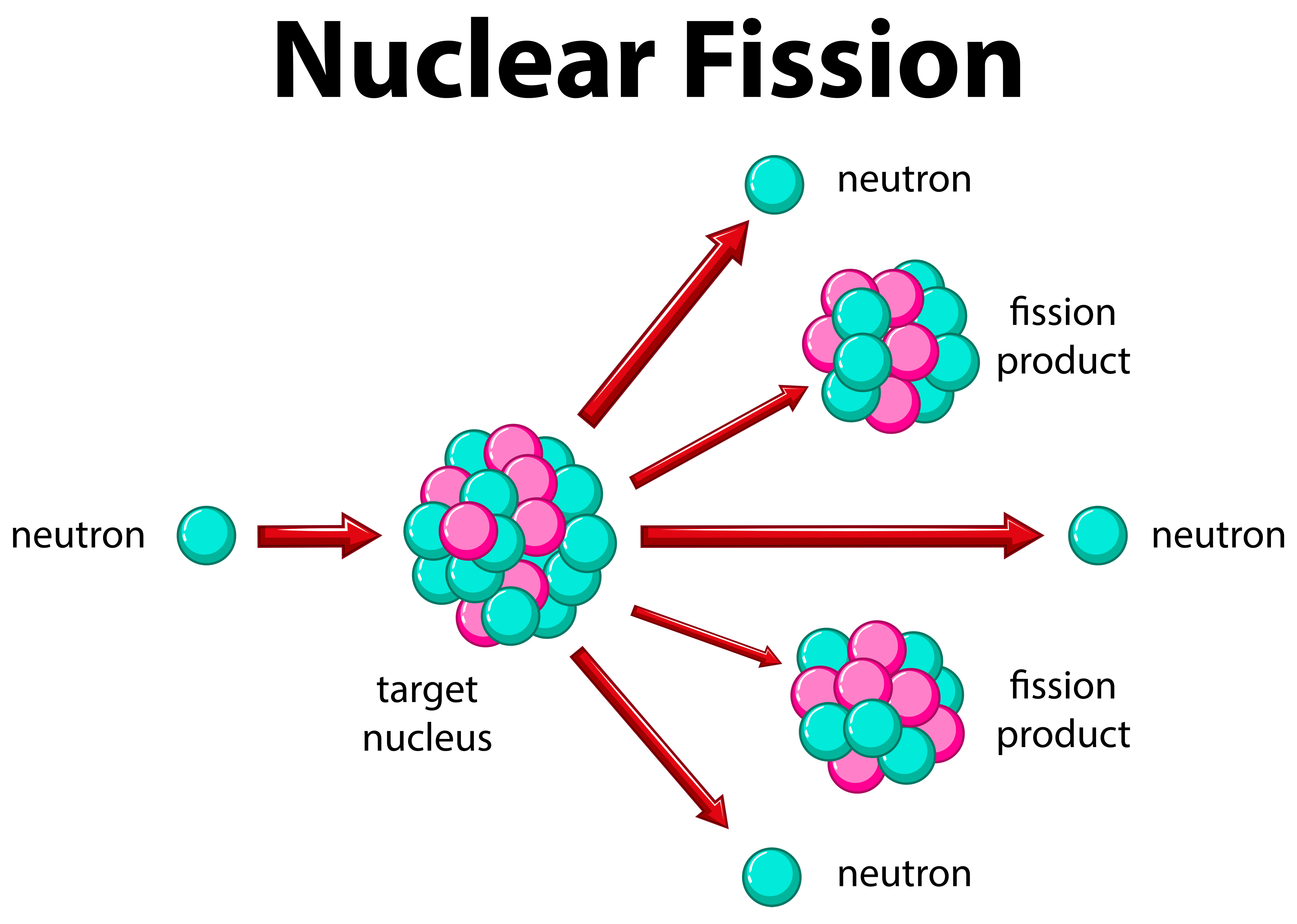
As such, the waste has to be housed for up to 10,000 years, cloistered from the environment and from anyone who might want to get at the plutonium for nefarious reasons. This waste fuel is highly radioactive and the culprits – these high-mass isotopes – have half-lives of many thousands of years. When the U235 content burns down to 0.3%, the fuel is spent, but it contains some very radioactive isotopes of americium, technetium, and iodine, as well as plutonium. When an atom of U238 absorbs a neutron, it transmutes into short-lived U239, which rapidly decays into neptunium-239 and then into plutonium-239, that lovely, weaponizable byproduct. The U238 does not just sit idly by, however it transmutes into other fissile elements. Once in the reactor, U235 starts splitting and releasing high-energy neutrons. 
As such, to make reactor fuel we have to expend considerable energy enriching yellowcake, to boost its proportion of U235. By contrast, the less-prevalent U235 is fissile. Most naturally occurring uranium is U238, but this common isotope does not undergo fission – which is the process whereby the nucleus splits and releases tremendous amounts of energy. The typical nuclear-fuel cycle starts with refined uranium ore, which is mostly U238 but contains 3% to 5% U235. In the post-Cold War world, is there any hope for thorium? Perhaps, but don't run to your broker just yet. The fact that thorium reactors could not produce fuel for nuclear weapons meant the better reactor fuel got short shrift, yet today we would love to be able to clearly differentiate a country's nuclear reactors from its weapons program. And here we come to it: Thorium reactors do not produce plutonium, which is what you need to make a nuke. The $2 billion Manhattan Project that produced the atomic bomb sparked a worldwide surge in nuclear research, most of it funded by governments embroiled in the Cold War. So why on earth are we using uranium? As you may recall, research into the mechanization of nuclear reactions was initially driven not by the desire to make energy, but by the desire to make bombs. And as proponents of the underdog fuel will happily tell you, thorium is more abundant in nature than uranium, is not fissile on its own (which means reactions can be stopped when necessary), produces waste products that are less radioactive, and generates more energy per ton. That means thorium could be used to fuel nuclear reactors, just like uranium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
